What is Paxlovid™?
In December 2021, the United States Food and Drug Administration (FDA) granted an Emergency Use Authorization (EUA) for
nirmatrelvir and ritonavir (Paxlovid™), which are separate pills packaged together and supposed to be taken together.
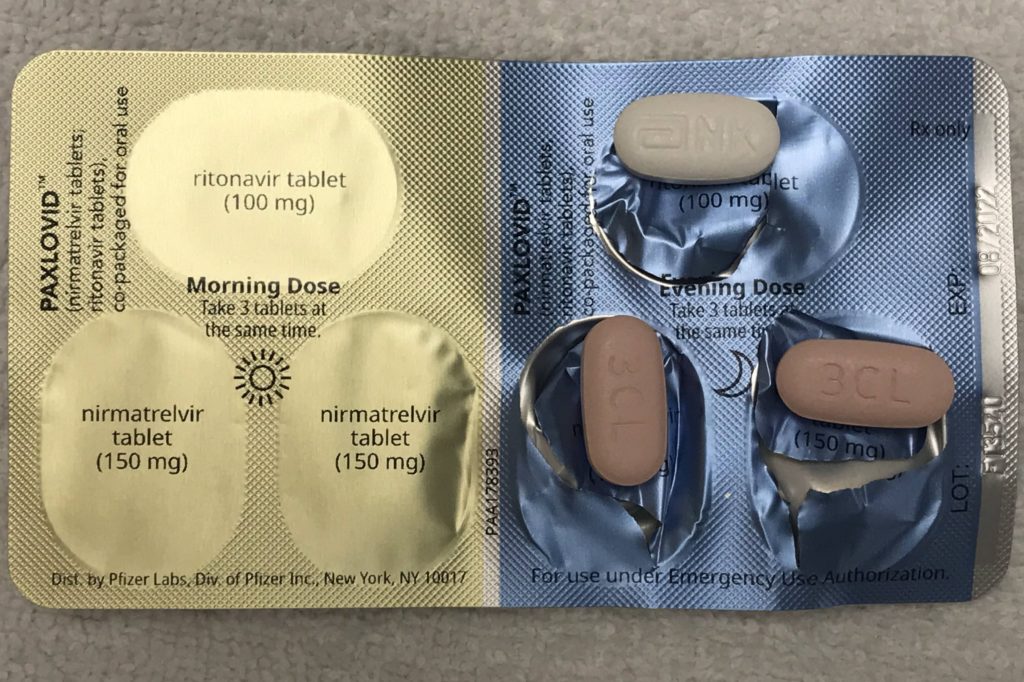
– Nirmatrelvir is a SARS-CoV-2 main protease inhibitor.
– Ritonavir was originally developed as an HIV-1 protease inhibitor but it is now used instead as a very strong inhibitor of cytochrome P450 3A4 (Ratain and Greenblatt., 2022). CYP3A, which refers to CYP3A4 and CYP3A5, is the most important enzyme for drug metabolism in humans (Ratain and Greenblatt., 2022). Ritonavir is the strongest clinically-available inhibitor of CYP3A (Ratain and Greenblatt., 2022). It is used to boost several antiviral medications that are used to treat HIV and hepatitis C (Ratain and Greenblatt., 2022).
If you are like me, you may find it hard to say “nirmatrelvir and ritonavir”, so, in the rest of this article, I’ll just use the abbreviation N+R, which has been used by other authors (Ratain and Greenblatt., 2022).
What is N+R authorized for?
The Emergency Use Authorization is for this combination of pills to be used for:
– The treatment of mild-to-moderate coronavirus disease 2019 (COVID-19)
– In patients who are at high risk for progression to severe COVID-19, including hospitalization or death.
Two clarifications:
1. The COVID-19 infection should have been confirmed by direct SARS-CoV-2 viral testing.
2. Regarding which age groups this authorization applies to, it is limited to:
– Adults
– Pediatric patients—who are a) 12 years of age and older, and b) weighing at least 40 kg
Where can patients get it?
Pharmacies and other locations where Paxlovid™ is available can be found at the following link: https://covid-19-therapeutics-locator-dhhs.hub.arcgis.com
Problem
Given that ritonavir is there because it inhibits the metabolic enzyme cytochrome P450 3A4, you can guess that this can lead to drug interactions with other medications that are metabolized by that enzyme.
A group of expert clinicians and antiviral pharmacologists from Europe and North America (Marzolini et al., 2022) predicted that the most important restriction to the use of N+R will be drug-drug interactions and the limited expertise of many clinicians regarding how to deal with them.
Another thing to remember is that we have to be ready to anticipate and manage drug interactions with N+R because patients will need N+R unexpectedly and urgently when they are diagnosed with COVID-19.
If a medication is metabolized by several pathways, of which CYP450 3A4 is only one, then the use of N+R is probably not going to be a big deal.
But, if CYP 3A4 is the main enzyme that metabolizes a particular medication, then, the levels of that medication can be expected to go up a lot, which is likely to cause adverse effects.
Inhibition of CYP3A4 occurs as soon as ritonavir is started and reaches its maximum in about 2 days (National Institutes of Health).
So, medications that are primarily metabolized by CYP3A4 should usually be stopped before N+R is started.
When the course of N+R is over, the inhibition of CYP 3A4 seems to go away relatively quickly. So, it has been recommended that the medication that had been stopped can usually be restarted 3 days after the course of N+R ends (Marzolini et al., 2022).
Which psychiatric medications does all this apply to? See below.
Enzyme inducers can lower levels of both nirmatrelvir and ritonavir
Some medications are potent inducers of cytochrome P450 enzymes. These medications can increase the metabolism of N+R and significantly lower its serum levels, which may be expected to reduce how well N+R works for that patient. So, this drug interaction is a big deal!
Among psychiatric medications, which ones can do this? Carbamazepine, St. John’s Wort. These two medications should not be given along with N+R.
And, it gets worse.
I said above that inhibition of CYP450 3A4 by ritonavir goes away after about 3 days. So, as discussed above, if the patient is on a medication that is primarily metabolized by CYP450 3A4 and it had to be stopped for the 5-day course of N+R, it can be restarted about 3 days after the course of Paxlovid.
But, enzyme induction (for example, by carbamazepine or St. John’s Wort) persists for a while even if we stop the carbamazepine or St. John’s Wort. So, if patients on carbamazepine/ St. John’s Wort get COVID-19 infection, unfortunately, the drug interaction cannot be avoided by simply stopping the carbamazepine/ St. John’s Wort. The patient will probably have to take something other than Paxlovid to treat the COVID-19 infection.
Clinical recommendations
1. Use an alternative COVID-19 treatment rather than N+R
– Some antipsychotics (because the serum levels of these medications may increase substantially): clozapine, lumateperone, lurasidone, pimozide
– Enzyme inducers (because they are likely to reduce the levels of N+R and, so, its efficacy): carbamazepine, St. John’s Wort
2. Temporarily stop the psychiatric medication
– For the following medications, we can consider stopping the medication if N+R is to be given and can restart it 3 days or more after the course of N+R ends.
– Some benzodiazepines: clonazepam, chlorazepate, diazepam, estazolam, flurazepam, triazolam
– Flibanserin
3. Consider reducing the dose; monitor carefully for possible side effects
Serum levels of the following medications are expected to increase. So, we should consider reducing the dose if N+R is to be started and monitor the patient for possible side effects.
– Alprazolam, chlordiazepoxide
– Aripiprazole, brexpiprazole, cariprazine, haloperidol, quetiapine, risperidone
– Buspirone
– Trazodone
– Zolpidem
4. Serum levels may DECREASE
Methadone: Serum levels of methadone may decrease by about one-third if N+R is started in a patient who is on methadone. This could lead to withdrawal symptoms. The decrease in methadone levels is due to the induction of its glucuronidation, which is a different mechanism from the inhibition of CYP 3A4 discussed above (Fact Sheet; COVID-19 Drug interaction Checker).
Recommended resource
The University of Liverpool’s COVID-19 Drug interactions website provides a PDF with color-coded guidance about drug interactions between selected medications and Paxlovid. A portion of that PDF is shown in the image below.
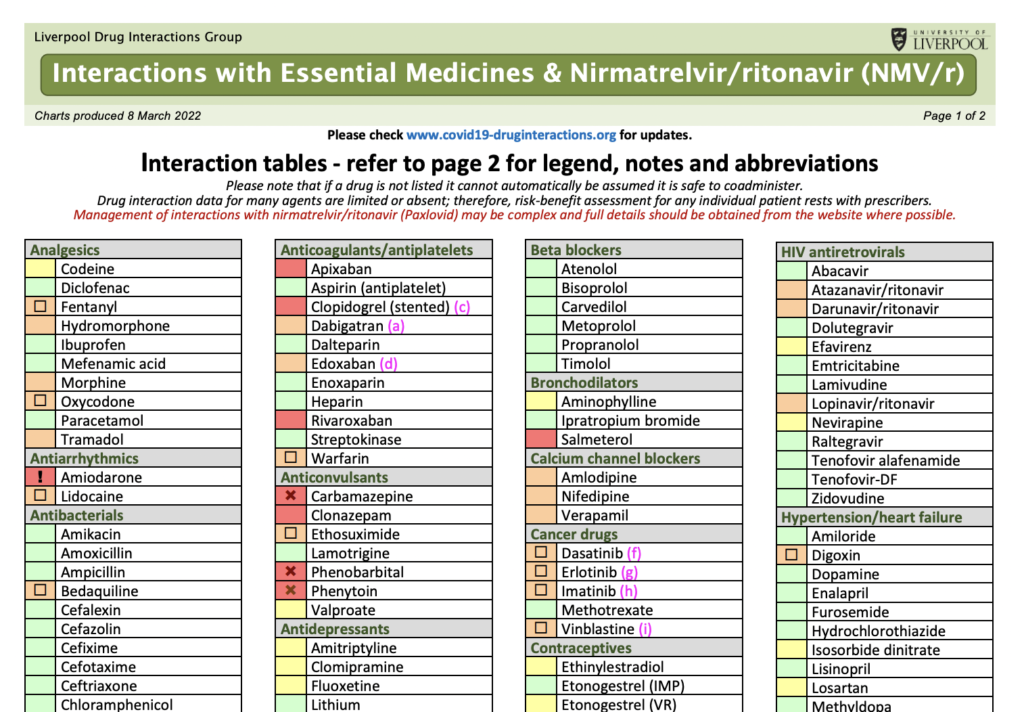
Next, please see the following article on this website:
COVID-19 drug interaction checker
External resources
COVID-19 Drug interactions (University of Liverpool)
Fact Sheet for Healthcare Providers: Emergency Use Authorization for Paxlovid™
National Institutes of Health. Ritonavir-Boosted Nirmatrelvir (Paxlovid)
Optional to read
Ethinyl estradiol (in oral contraceptives). N+R may decrease ethinyl estradiol concentrations (Fact Sheet). So, the official recommendation is that “An additional, non-hormonal method of contraception should be considered during the 5 days of PAXLOVID treatment and until one menstrual cycle after stopping PAXLOVID” (Fact Sheet). But, it has been pointed out that (National Institutes of Health):
– Since N+R is given for only 5 days, this would not be expected to decrease the effectiveness of combination oral contraceptives
– Also, combination oral contraceptives containing ethinyl estradiol also contain a progestin, whose concentration is not expected to decrease.
Buprenorphine: Though ritonavir increases exposure to buprenorphine and its active metabolite, this did not lead to clinically significant problems. So, this is considered a weak drug interaction.
Related Pages
A simple guide to the COVID-19 vaccines
A primer on tests for identifying 2019 Coronavirus (SARS-CoV-2) infection
Important recommendations regarding sex and coronavirus
What mental health clinicians can do about the coronavirus
Could psychotropic medications affect the immune response to coronavirus?
Helping persons with OCD deal with the coronavirus pandemic
The relationship between COVID-19 and mental disorders (and what we should do about it)
Mental disorders and the immune system
Cytochrome P450 system and psychiatric medications: An introduction and important tips
Cytochrome P450 3A4: Substrates, inhibitors, and inducers
The Flockhart P450 Drug Interaction Table: Full table (Link to external site)
Supplement to Flockhart table
Antidepressants with much fewer P450 drug interactions
One exit door or many?
References
COVID-19 Drug interactions (University of Liverpool)
Fact Sheet for Healthcare Providers: Emergency Use Authorization for Paxlovid™
National Institutes of Health. Ritonavir-Boosted Nirmatrelvir (Paxlovid)
Girardin F, Manuel O, Marzolini C, Buclin T. Evaluating the risk of drug-drug interactions with pharmacokinetic boosters: the case of ritonavir-enhanced nirmatrelvir to prevent severe COVID-19. Clin Microbiol Infect. 2022 Mar 28:S1198-743X(22)00169-0. doi: 10.1016/j.cmi.2022.03.030. Epub ahead of print. PMID: 35358684; PMCID: PMC8958820.
Marzolini C, Kuritzkes DR, Marra F, Boyle A, Gibbons S, Flexner C, Pozniak A, Boffito M, Waters L, Burger D, Back D, Khoo S. Prescribing Nirmatrelvir-Ritonavir: How to Recognize and Manage Drug-Drug Interactions. Ann Intern Med. 2022 Mar 1:M22-0281. doi: 10.7326/M22-0281. Epub ahead of print. PMID: 35226530; PMCID: PMC8890619.
Ratain MJ, Greenblatt DJ. Drug Interactions with a Short Course of Nirmatrelvir and Ritonavir: Prescribers and Patients Beware. J Clin Pharmacol. 2022 Apr 12. doi: 10.1002/jcph.2060. Epub ahead of print. PMID: 35412678.
S K SR, P A A, B S, Kalala KP, Pm A, Sabarathinam S. Drug interaction risk between cardioprotective drugs and drugs used in treatment of COVID-19: A evidence-based review from six databases. Diabetes Metab Syndr. 2022 Mar;16(3):102451. doi: 10.1016/j.dsx.2022.102451. Epub 2022 Mar 7. PMID: 35279008; PMCID: PMC8898923.
Copyright © 2025 to 2026, Simple and Practical Medical Education, LLC. All rights reserved. The content on this website may not be reproduced in any form without express written permission.
Disclaimer: The material on this website is provided as general education for medical professionals. It is not intended for patients or other laypersons, nor is it a substitute for medical advice, diagnosis, or treatment. Patients must always consult a qualified healthcare professional regarding their diagnosis and treatment. Healthcare professionals should always check this website for the most up-to-date information.
Excellent and perfect timing! Thank you!!!!
To avoid benzo withdrawal during stoppage ie with clonazepam, might it make sense to switch to lorazepam temporarily which doesn’t seem to have significant interactions? I would not be concerned about stopping diazepam for 5 + 3 days but shorter acting might get quite uncomfortable